SOLVED: A 35-g ice cube at 0.0 °C is added to 110 g of water in a 62-g aluminum cup. The cup and the water have an initial temperature of 23 °C. (
By A Mystery Man Writer
Description
VIDEO ANSWER: Hello students to solve the given question: let us use the equation of heat transfer that is equal to m c c. Here is the specific heat capacity multiplied by delta t that is, temperature difference now, using this relation? Let us solve
Numerade is a venture-backed, high-growth education technology startup based in Pasadena. We are singularly focused on creating exceptional video and interactive content experiences for education making the knowledge and skills of world class educators widely accessible and affordable to student audiences of all backgrounds. Our mission is to close the educational opportunity gap by unlocking and democratizing access to extraordinary educators and the content they have to offer.
Numerade is a venture-backed, high-growth education technology startup based in Pasadena. We are singularly focused on creating exceptional video and interactive content experiences for education making the knowledge and skills of world class educators widely accessible and affordable to student audiences of all backgrounds. Our mission is to close the educational opportunity gap by unlocking and democratizing access to extraordinary educators and the content they have to offer.
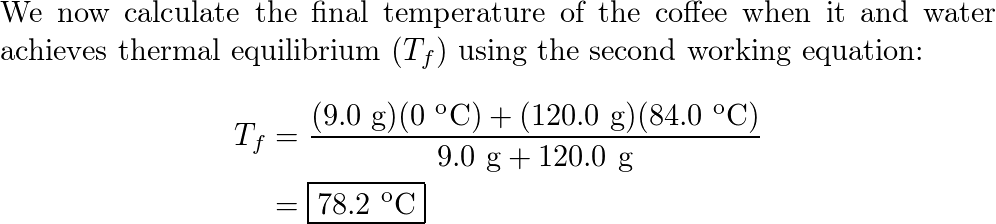
An ice cube of mass 9.0 g is added to a cup of coffee. The c

Answered: Two 20.0 g ice cubes at −13.0 ∘C are…
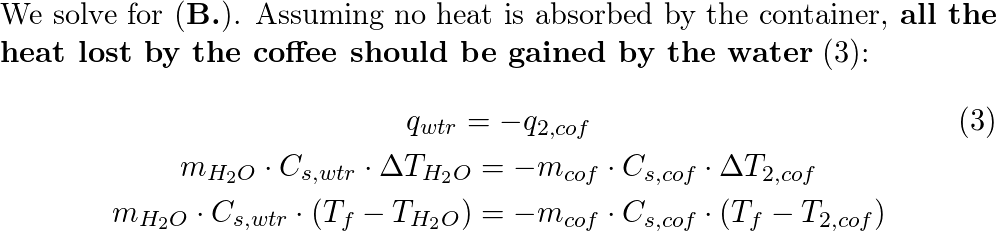
An ice cube of mass 9.0 g is added to a cup of coffee. The c

Specific Heat Capacity
10g of ice cubes at 0 degree Celsius is released in a tumbler

Finding Final Temperature When Ice is Added to Water

Answered: A 100g cube of ice at 0°C is dropped…
200 grams of water at 90°C is mixed with 100 gram of water at 30°C
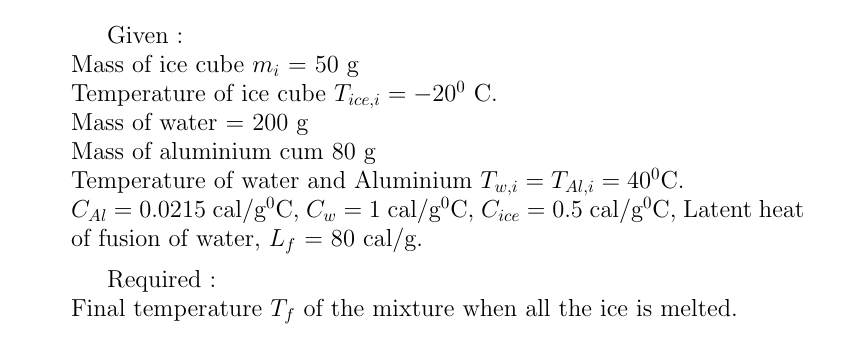
Answered: A 50 g ice cube, initially at -20degree…
from
per adult (price varies by group size)