32. 80 g of h2 is reacted with 80 g of o2 to form water. find out the mass of water obtained.which substance is the limiting reagent.
By A Mystery Man Writer
Description
32. 80 g of h2 is reacted with 80 g of o2 to form water. find out the mass of water obtained.which substance is the limiting reagent.
32- 80 g of h2 is reacted with 80 g of o2 to form water- find out the mass of water obtained-which substance is the limiting reagent
32- 80 g of h2 is reacted with 80 g of o2 to form water- find out the mass of water obtained-which substance is the limiting reagent

SOLVED: Which is the limiting reactant when 5.00 g of H2 and 10.0 g of O2 react and form water? [a] How much water is produced? [b] How much excess reagent remains? [

80 gram of H2 is reacted with 80 gram of O2 to form water find out the mass of water obtained which

Gas Stoichiometry - Chemistry
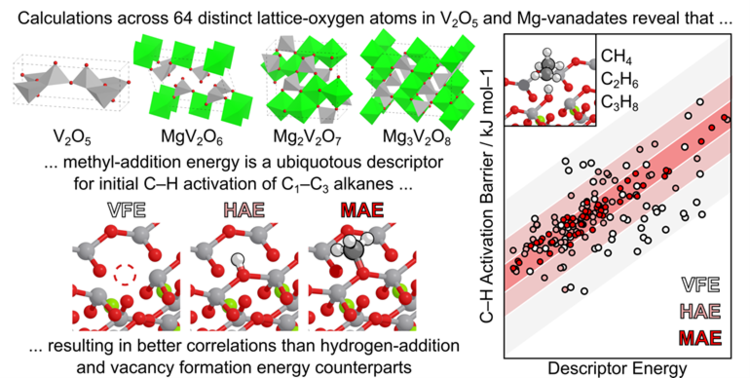
Hibbitts Group Publications

PS GR 11 Session 11 LN

PPT - Mass Relationships in Chemical Reactions PowerPoint Presentation - ID:3181342
Hydrogen and oxygen react chemically to form water. How much

Answered: A reaction vessel contains14.1 g of CO…

What volume (in liters) of water vapor will be produced when 26 L of H2 and 13 L of O2 react according to the equation given below? (Assume that the reactants and
Oxygen, Free Full-Text

Answered: 3. Hydrogen and oxygen gas combine to…

Direct production of H2O2 from H2 and O2 in a biphasic H2O/scCO2 system over a Pd/C catalyst: Optimization of reaction conditions - ScienceDirect

US10328082B2 - Methods of use and combinations - Google Patents
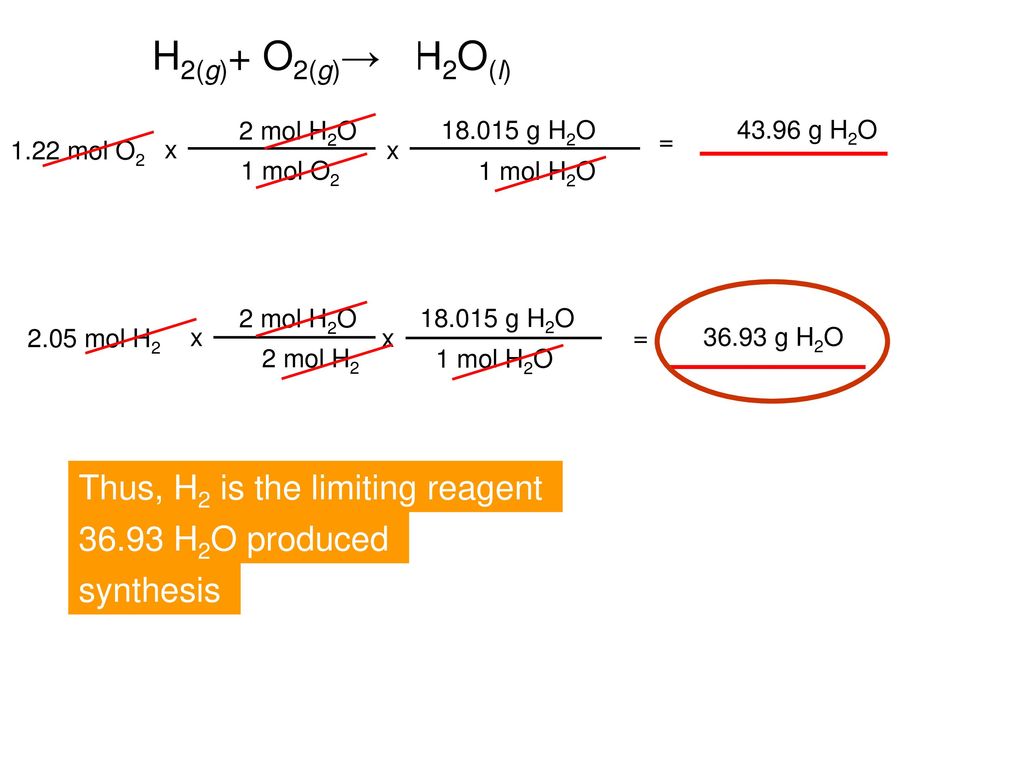
2H2(g)+ O2(g)→ 2H2O(l) Thus, H2 is the limiting reagent - ppt download
from
per adult (price varies by group size)






