Class II Device Definition
By A Mystery Man Writer
Description
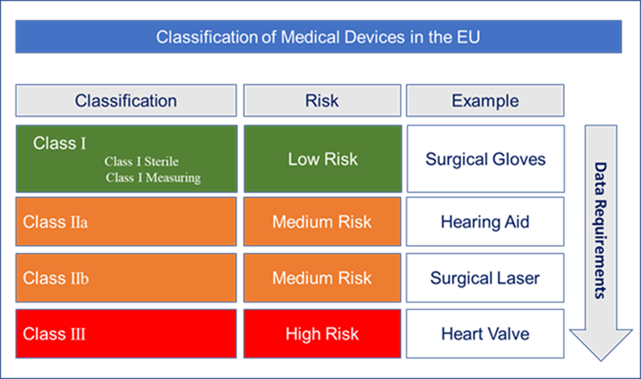
Class II medical devices have moderate to higher risks to patients or users. Over 40% of medical devices fall into this device category. The majority of medical devices are considered to be Class II devices. Some examples of Class II devices include catheters, syringes, contact lens, and pregnancy test kits.
Approaching MDR Compliance
How to Classify Your Medical Device for FDA Approval

Medical device regulations, classification & submissions

Entering the US Market: Medical Devices - ppt video online download

Japan Health Policy NOW – 6.4 Medical Devices

What's the Difference between a Class I Medical Device and a Class II?

SOCRA CCRP Exam - Term: Definition: What is in 21 CFR Part 11

Guidance Document - Guidance on the Risk-based Classification System for Non-In Vitro Diagnostic Devices (non-IVDDs)
Classify Your Medical Device
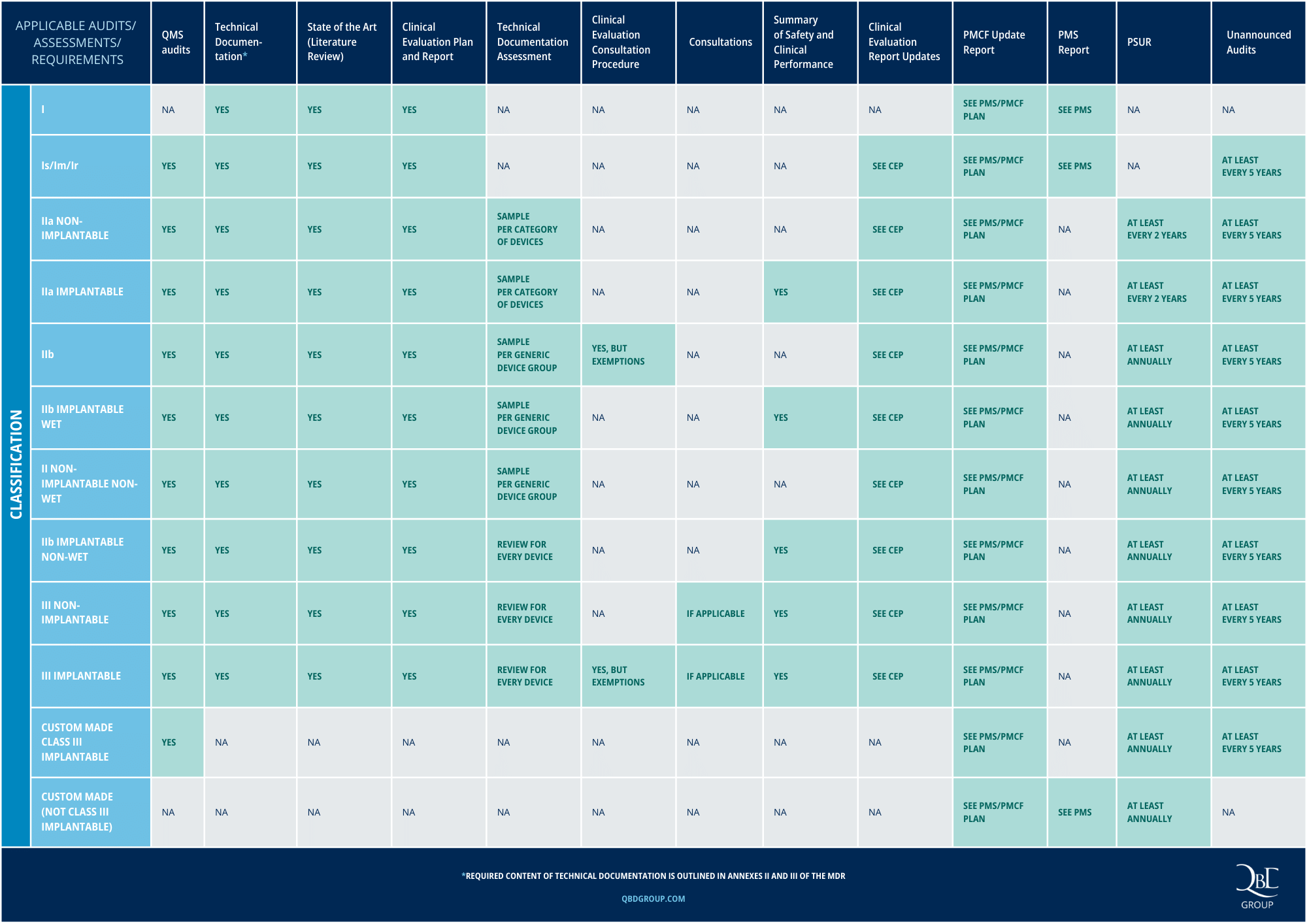
CE Approval for Medical Devices under MDR: key requirements per device class
from
per adult (price varies by group size)