Microbial Culture Media For Quality Control Of Non-Sterile Products
By A Mystery Man Writer
Description
lt;p>Using the correct media is critical to ensure microbiological quality. Explore a portfolio of culture media and substances for sample preparation, microbial enumeration tests, and tests for specified microorganisms.</p>
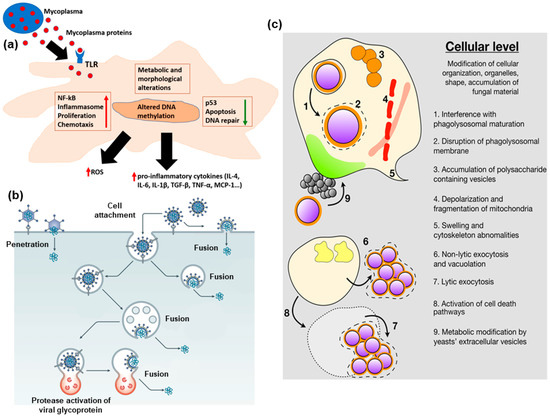
Chemosensors, Free Full-Text

Microbiological testing for medical devices
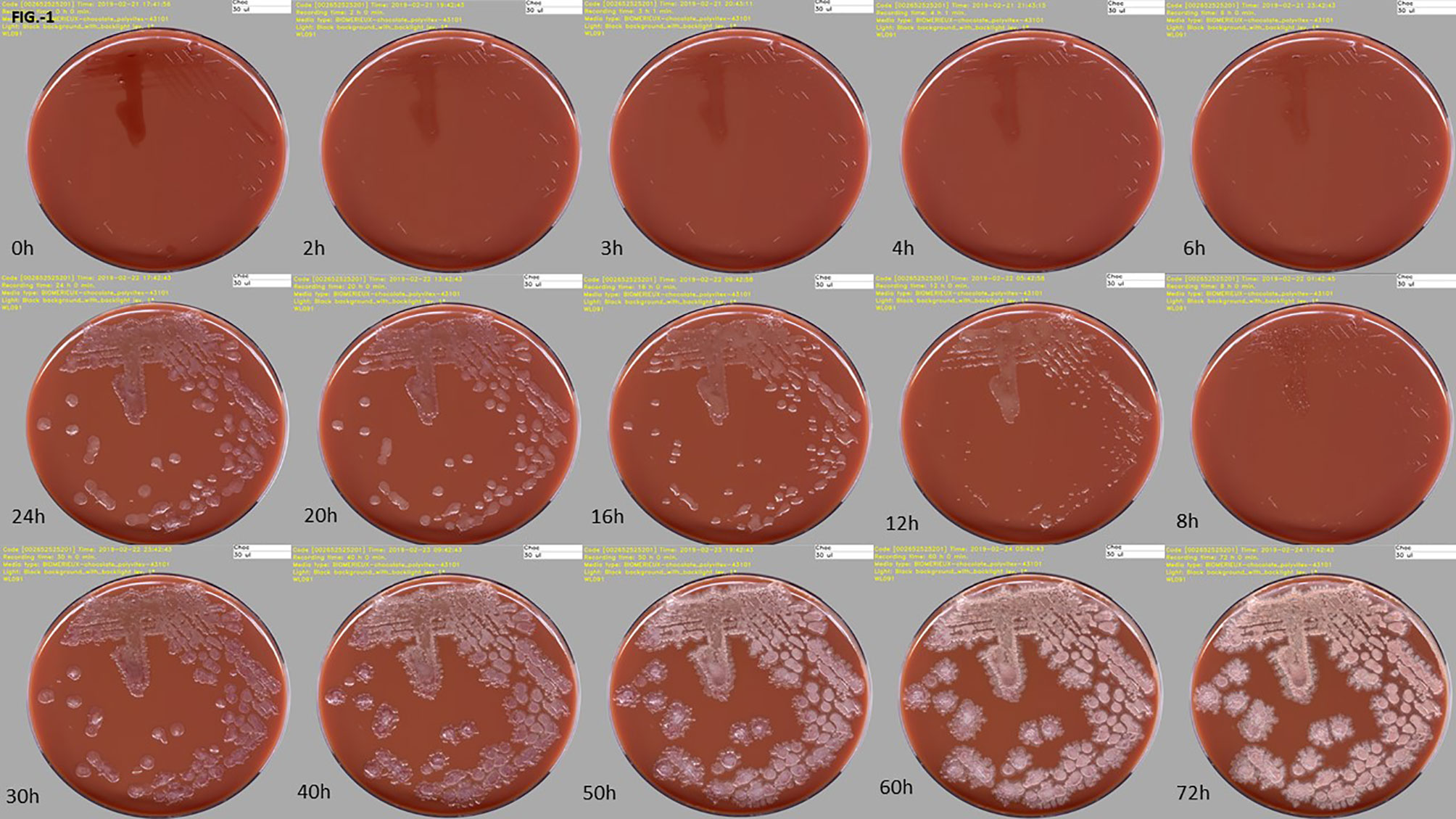
Frontiers Total Laboratory Automation for Rapid Detection and Identification of Microorganisms and Their Antimicrobial Resistance Profiles
Microbiology Series: Water Activity

Pharmaceutical Microbiology Thermo Fisher Scientific - UK
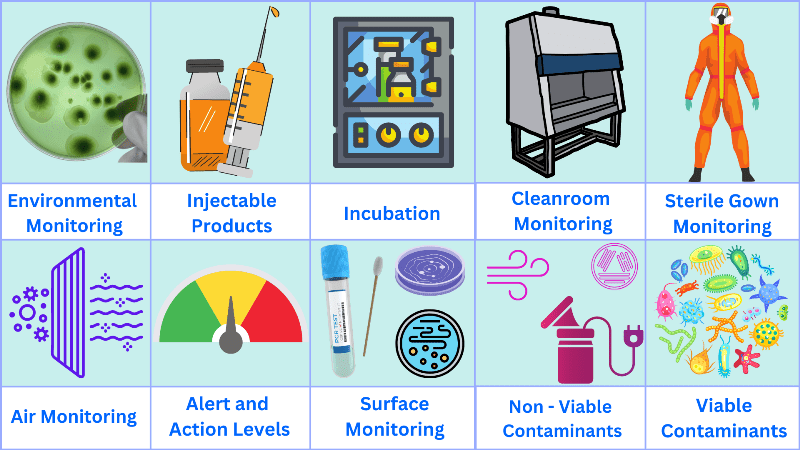
What is environmental monitoring in pharmaceutical industry
Biologics Quality Control A Critical Component Of Development And Production

Quality Control for Microbiological Culture Media - ppt video online download
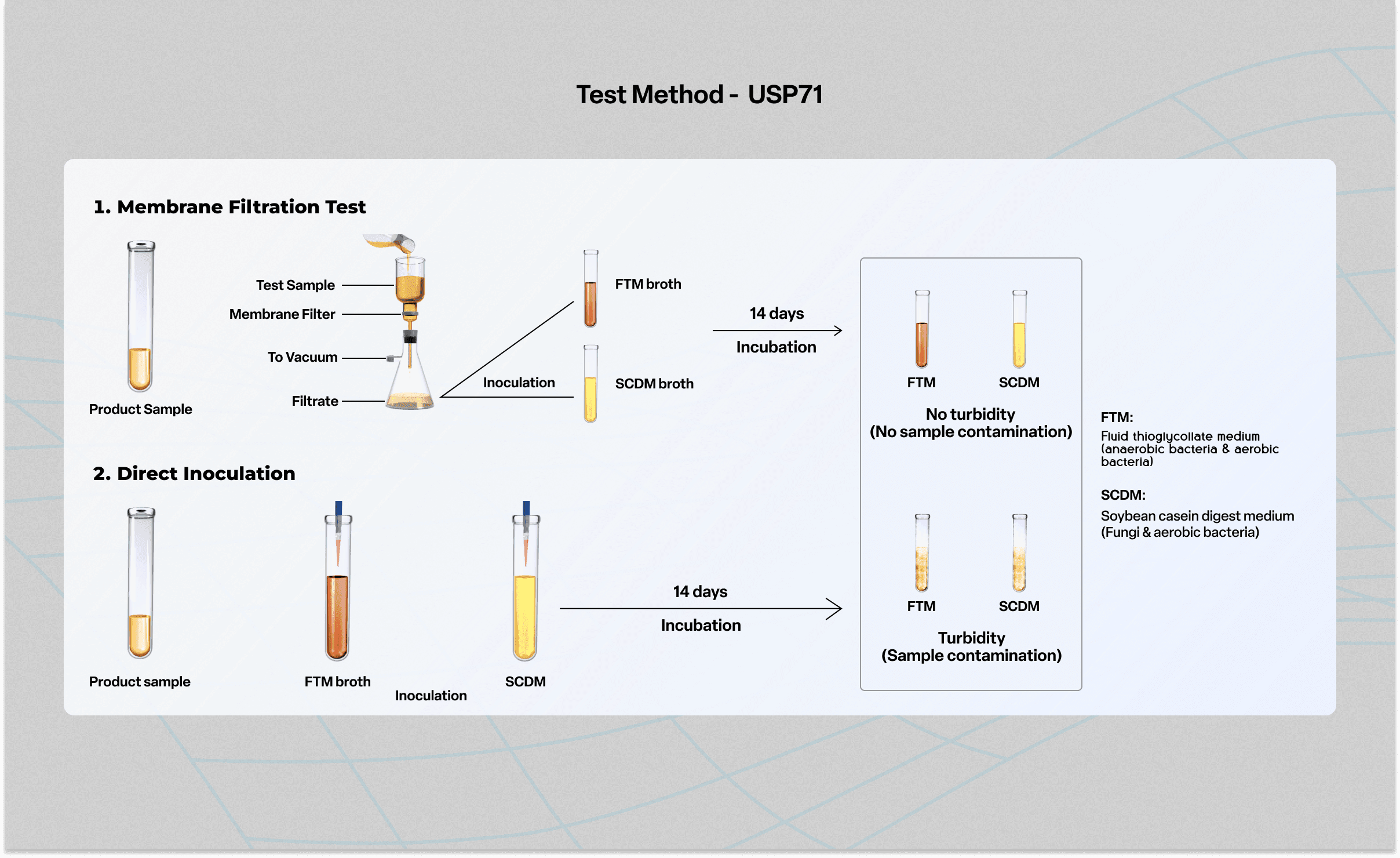
USP 71 - Sterility Testing of Pharmaceutical Products.

Microbial Enumeration for Non-Sterile Products
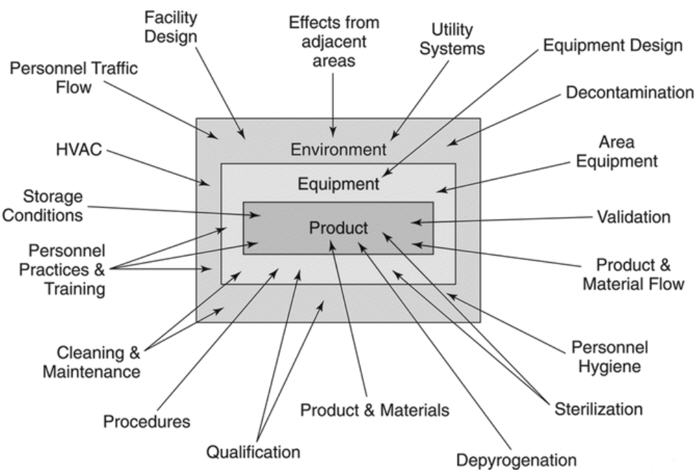
The Essential Components Of A Sterility Assurance Program

vertassets.blob.core.windows.net/image/17ccca9b/17

Microbiology Testing for Non-sterile Products

QUALITY CONTROL OF PARENTERALS,STERILE PRODUCT
High Complexity Media-Fill Test Kit - IVQA
from
per adult (price varies by group size)







