Low coverage whole genome sequencing enables accurate assessment
By A Mystery Man Writer
Description
Background Inherited susceptibility to common, complex diseases may be caused by rare, pathogenic variants (“monogenic”) or by the cumulative effect of numerous common variants (“polygenic”). Comprehensive genome interpretation should enable assessment for both monogenic and polygenic components of inherited risk. The traditional approach requires two distinct genetic testing technologies—high coverage sequencing of known genes to detect monogenic variants and a genome-wide genotyping array followed by imputation to calculate genome-wide polygenic scores (GPSs). We assessed the feasibility and accuracy of using low coverage whole genome sequencing (lcWGS) as an alternative to genotyping arrays to calculate GPSs. Methods First, we performed downsampling and imputation of WGS data from ten individuals to assess concordance with known genotypes. Second, we assessed the correlation between GPSs for 3 common diseases—coronary artery disease (CAD), breast cancer (BC), and atrial fibrillation (AF)—calculated using lcWGS and genotyping array in 184 samples. Third, we assessed concordance of lcWGS-based genotype calls and GPS calculation in 120 individuals with known genotypes, selected to reflect diverse ancestral backgrounds. Fourth, we assessed the relationship between GPSs calculated using lcWGS and disease phenotypes in a cohort of 11,502 individuals of European ancestry. Results We found imputation accuracy r2 values of greater than 0.90 for all ten samples—including those of African and Ashkenazi Jewish ancestry—with lcWGS data at 0.5×. GPSs calculated using lcWGS and genotyping array followed by imputation in 184 individuals were highly correlated for each of the 3 common diseases (r2 = 0.93–0.97) with similar score distributions. Using lcWGS data from 120 individuals of diverse ancestral backgrounds, we found similar results with respect to imputation accuracy and GPS correlations. Finally, we calculated GPSs for CAD, BC, and AF using lcWGS in 11,502 individuals of European ancestry, confirming odds ratios per standard deviation increment ranging 1.28 to 1.59, consistent with previous studies. Conclusions lcWGS is an alternative technology to genotyping arrays for common genetic variant assessment and GPS calculation. lcWGS provides comparable imputation accuracy while also overcoming the ascertainment bias inherent to variant selection in genotyping array design.

Systematic dissection of biases in whole-exome and whole-genome sequencing reveals major determinants of coding sequence coverage

How a Cutting-Edge Genetic Test Helps Ill Newborns, Pregnant Moms

SciELO - Brasil - Clinical genomics and precision medicine Clinical genomics and precision medicine

Identification of RP1 as the genetic cause of retinitis pigmentosa in a multi-generational pedigree using Extremely Low-Coverage Whole Genome Sequencing (XLC-WGS) - ScienceDirect
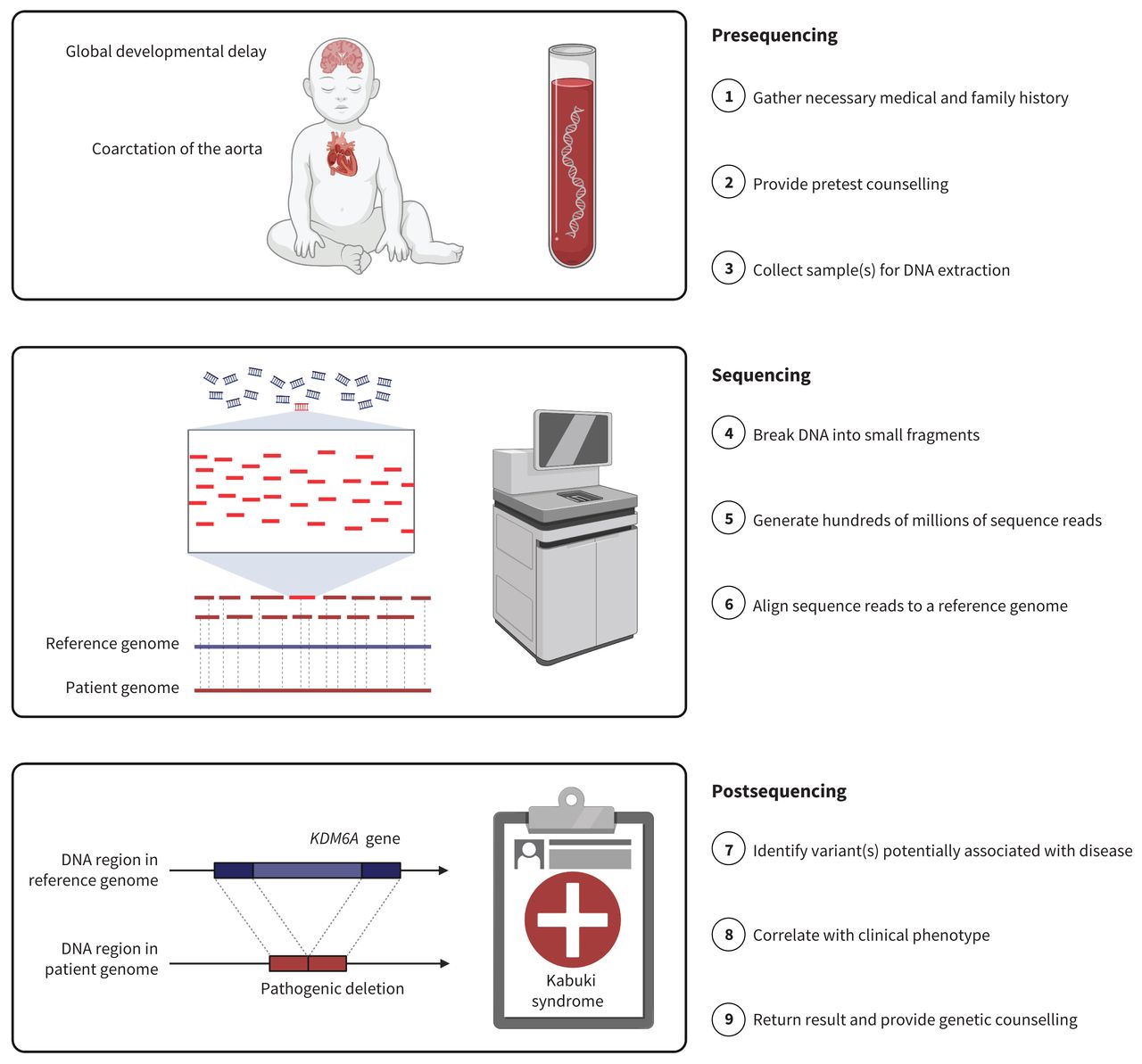
Genome sequencing as a diagnostic test
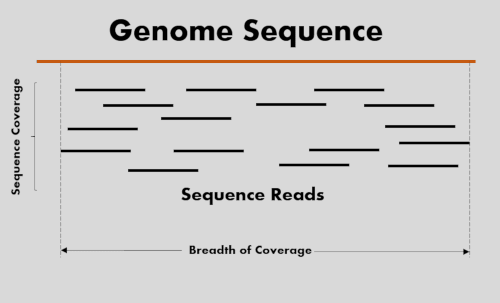
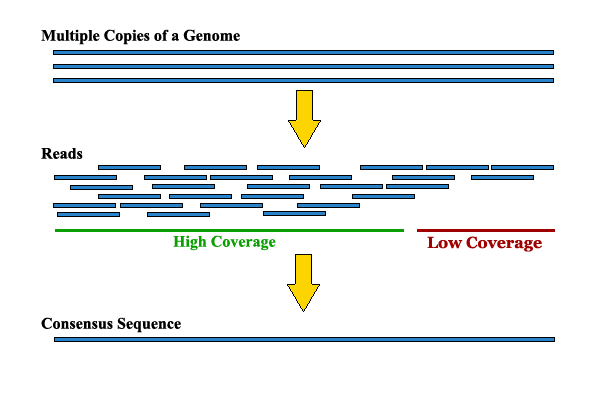
Sequencing coverage and breadth of coverage

Frontiers Ultra Low-Coverage Whole-Genome Sequencing as an Alternative to Genotyping Arrays in Genome-Wide Association Studies

Metagenomics next-generation sequencing tests take the stage in the diagnosis of lower respiratory tract infections - ScienceDirect
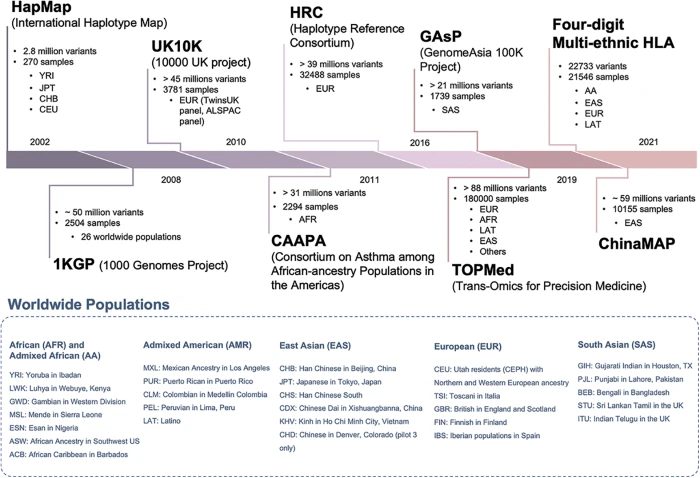
BIOCELL, Free Full-Text
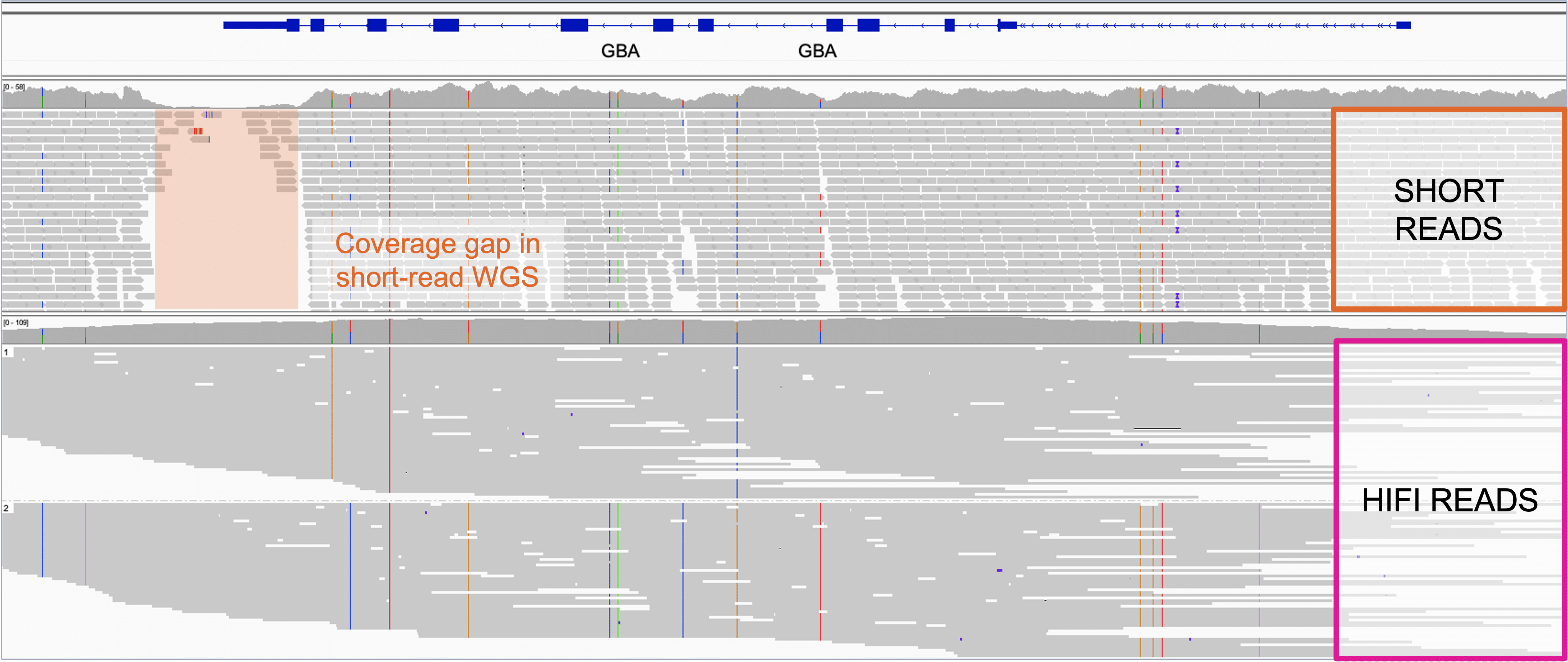
Sequencing 101: Sequencing coverage - PacBio
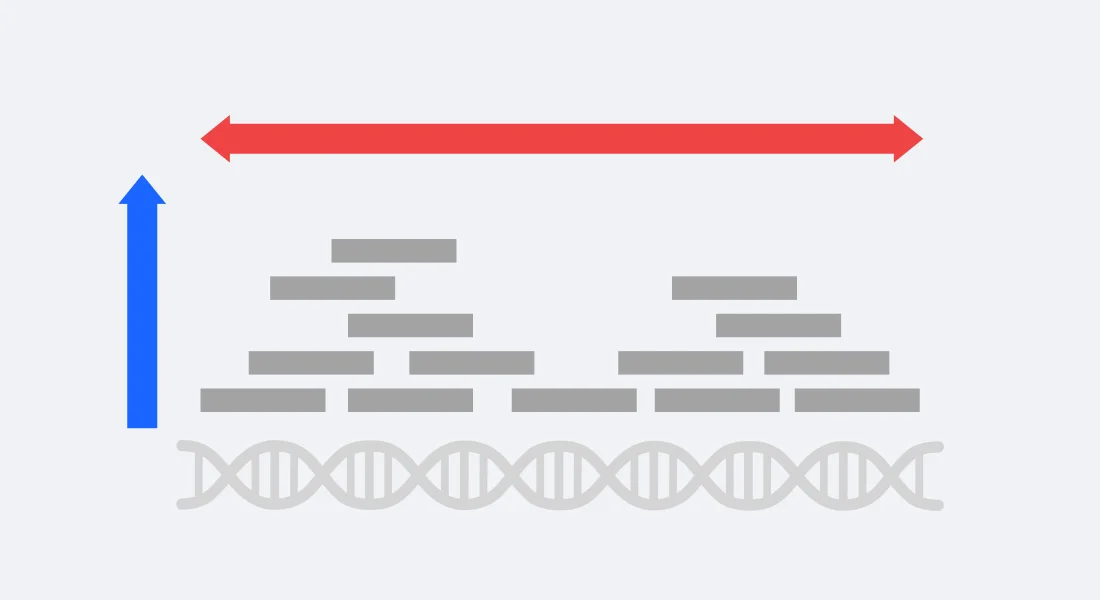
Sequencing Depth vs Coverage

Low-Pass Genome Sequencing: Validation and Diagnostic Utility from 409 Clinical Cases of Low-Pass Genome Sequencing for the Detection of Copy Number Variants to Replace Constitutional Microarray - ScienceDirect

Relative matching using low coverage sequencing
from
per adult (price varies by group size)