Applications for Medical Device Investigational Testing Authorizations Guidance Document
By A Mystery Man Writer
Description
Applications for Medical Device Investigational Testing Authorizations Guidance Document

Applications for Medical Device Investigational Testing Authorizations Guidance Document

What Labs Need to Know About New Medical Device Cybersecurity Rules
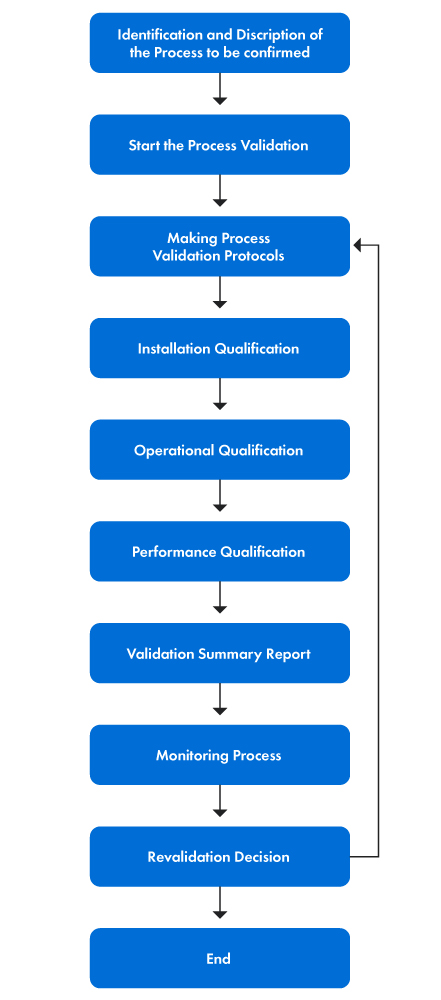
Steps involved in US FDA's Medical devices Validation Protocol Process
List of documents to upload to the Spanish Agency of Medicines and
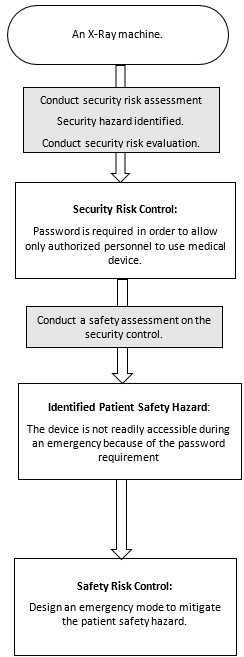
Guidance Document: Pre-market Requirements for Medical Device

Class II - IV Medical Device Investigational Testing in Canada - Vantage BioTrials

Frontiers A Regulatory Risk-Based Approach to ATMP/CGT Development: Integrating Scientific Challenges With Current Regulatory Expectations

Guidance on how to complete the application for a new medical device licence: Overview

Necessity of strengthening the current clinical regulatory for companion diagnostics: An institutional comparison of the FDA, EMA, and MFDS - ScienceDirect

Medical Device Resources & Webinars from Greenlight Guru

Health Canada medical device regulations
Clinical Research with Medical Devices 101

Health Canada guidance on anonymization and redaction of clinical data
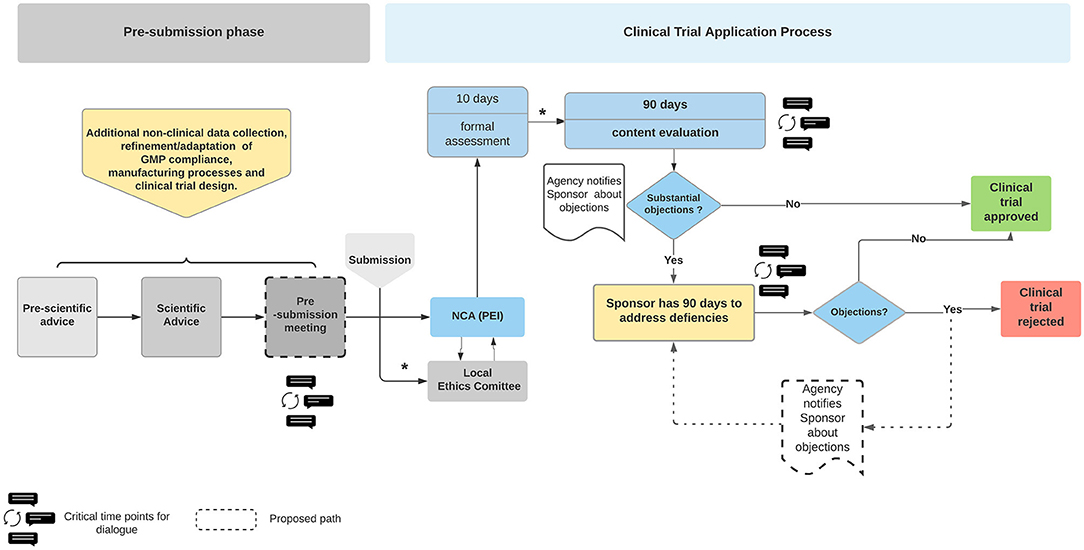
Frontiers Advanced Therapy Medicinal Products' Translation in Europe: A Developers' Perspective
from
per adult (price varies by group size)







