Five Common Mistakes Submitting a Premarket Notification
By A Mystery Man Writer
Description
How you can avoid the most common errors made when submitting a 510(k), the “premarket notification,” with simple measures

Common Mistakes in Safety Analytics for FDA Submissions

Premarket Notification The 510(k) Process
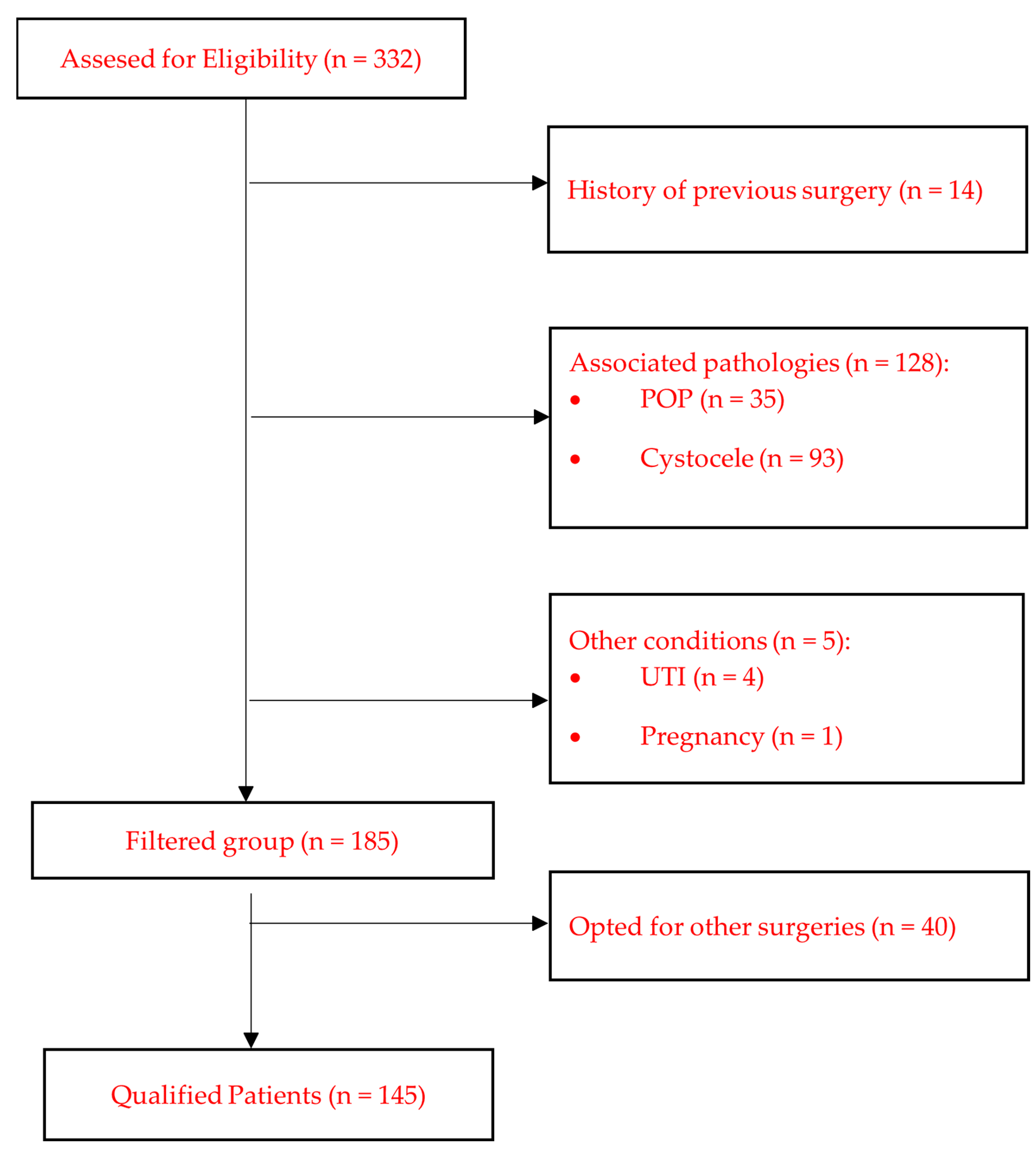
Life, Free Full-Text

CRSToday The Journey From Bench to Market

Robert A. Allen, PhD on LinkedIn: #biocompatibility #meddevice #medicaldevice #medicaldevices…

FDA 21 CFR Part 820: 8 Most Common Mistakes to Avoid

FDA Releases New Cybersecurity Premarket Guidance

Key Considerations for Successful Creation and Submission of a 510(k) eCopy

FDA 510(k) Submission: A Step-By-Step Guide On How To Prepare Yours

Premarket Notification The 510(k) Process

The Top 10 Most Significant Changes Introduced by the New EU MDR (and how to avoid the common mistakes)

Mock FDA 510(k) Filing

Dow closes 300 points lower after strong jobs data signals more rate hikes
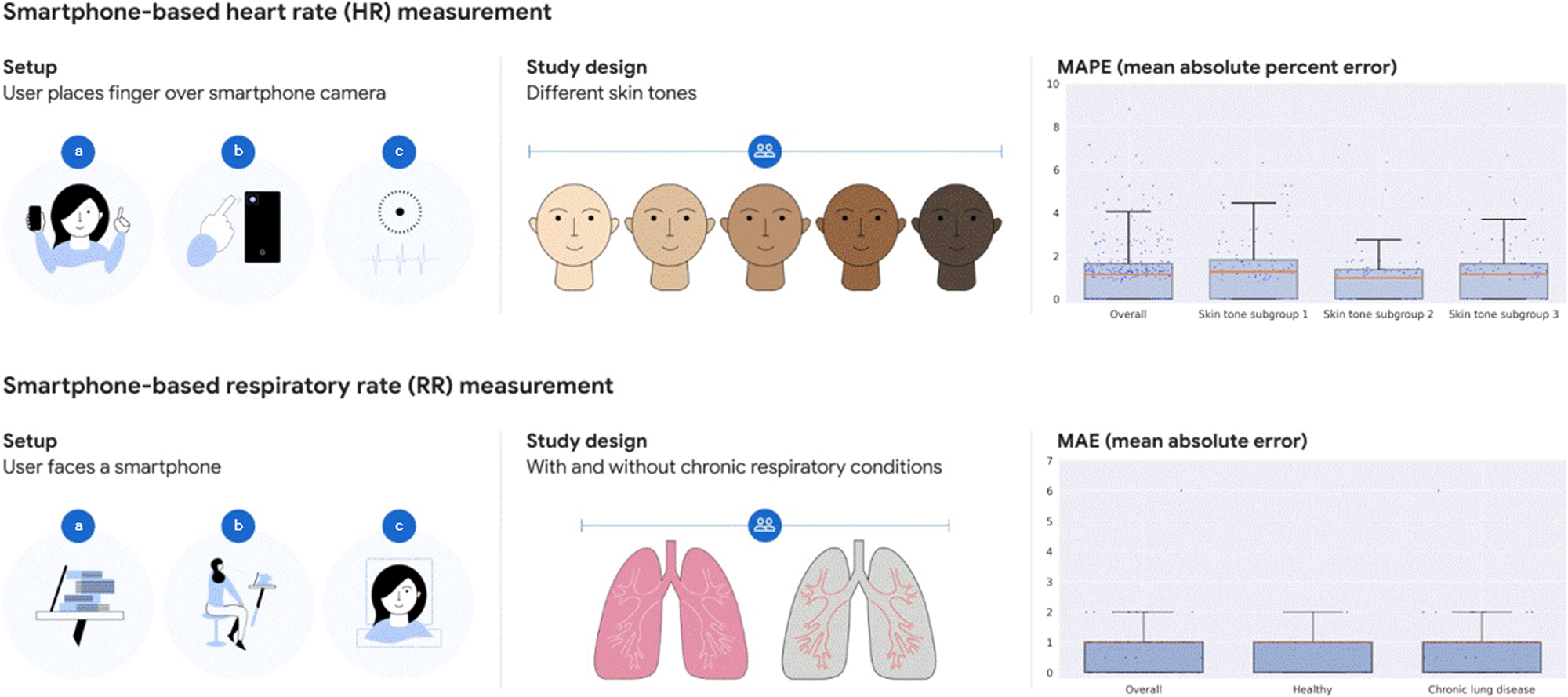
Prospective validation of smartphone-based heart rate and respiratory rate measurement algorithms
from
per adult (price varies by group size)







