42g of N₂ react with excess of O₂ to produce NO. Amount of NO formed is a.60g b.32g c.45g d.90g
By A Mystery Man Writer
Description
Share your videos with friends, family and the world

5.2: Reaction Stoichiometry (Problems) - Chemistry LibreTexts
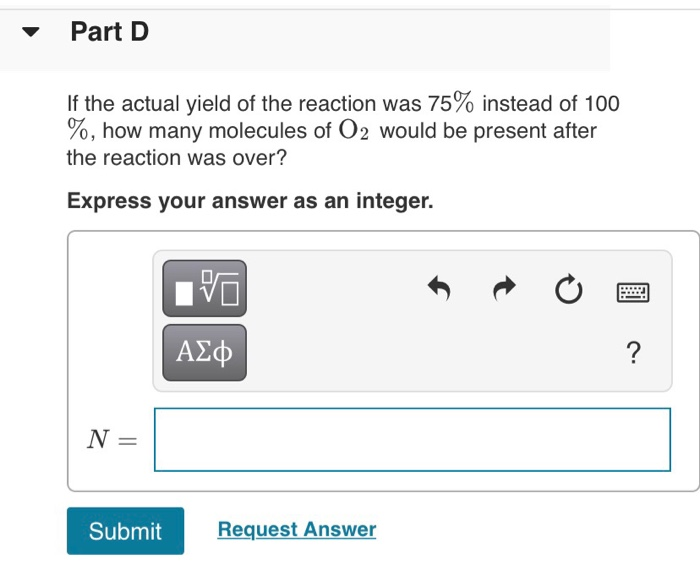
Solved Nitrogen monoxide and oxygen react to form nitrogen

JP2009534398A - Cyclohexylpyrazole-lactam derivatives as

5.2: Reaction Stoichiometry (Problems) - Chemistry LibreTexts

WO2022045231A1 - Ester compound - Google Patents

Topical Mock Chemistry Questions, PDF

Chem Questions by Topic, PDF, Chemical Elements

UMAIR KHAN ACADEMY

Chem Questions by Topic, PDF, Chemical Elements

WO2022045231A1 - Ester compound - Google Patents

Topical Mock Chemistry Questions, PDF, Chemical Elements

UMAIR KHAN ACADEMY

Percent Yield Calculator

Topical Mock Chemistry Questions, PDF
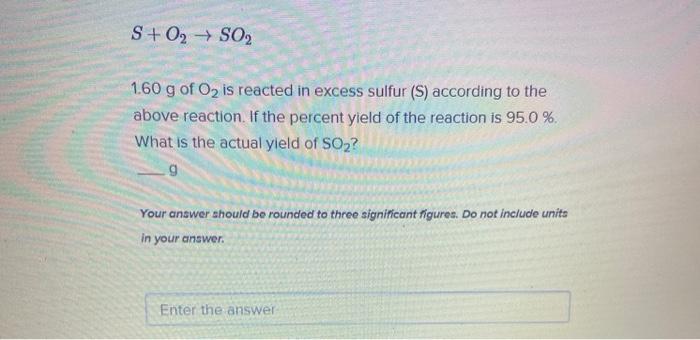
Solved S + O2 + S02 1.60 g of O2 is reacted in excess sulfur
from
per adult (price varies by group size)







