For a given gas, a graph is shown between compressibility factor (Z) and Pressure (P).Select the incorrect statement(s) about the various temperature relations.a)Temperature T1 must be above critical temperature (TC).b)Temperature T2 may
By A Mystery Man Writer
Description

Compressibility factor Z is plotted against pressure p for four different gases A , B , C & D. The correct order of critical temperature of the gasesA. A>B>C>DB. B>A>C>DC. D

Compressibility Factor Z Important Concepts and Tips for JEE Main
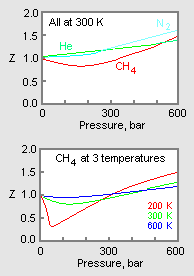
Compressibility factor (gases) - Knowino

Compressibility Factor - an overview

PV Compressibility factor Z= nRT is plotted against pressure : N. Ideal gas What is the correct order of liquefiability of the gases shown in the above graph? H

A graph Z vs P is plotted N_2 gas different temperatureThe correct relationship between temperatures

The following graph is plotted between compressibility factor Z versus pressure of a gas at different temperatures.Which of the following statements is /are correct?

The graph of compressibility factor (Z) v/s P 1 mol of a real gas is shown in following diagram. The graph is plotted 273 K temperature. If slope of graph very high

The given graph represents the variation of compressibility factor Z vs P for three gases A, B and C.Identify the incorrect statements.
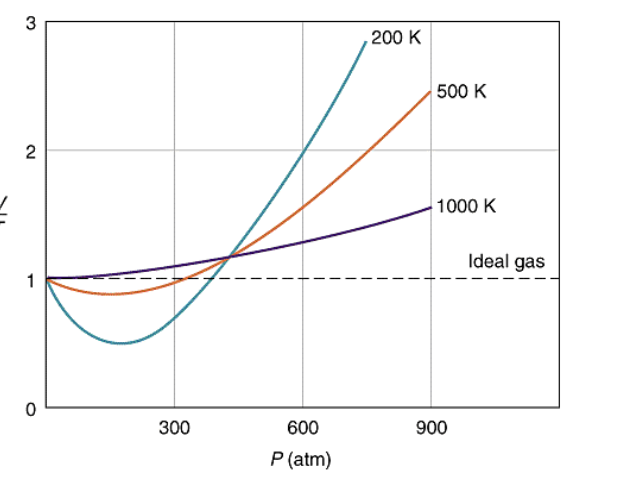
Solved 1. The plot below shows how compressibility factor

compressibility Factor v/s Pressure/ Temperature Graph . States of Matter Class XI.

Temperature and Pressure Measurements of an Ideal Gas - Because the ideal gas was in a closed - Studocu

Compressibility factor (Z=(PV)/(nRT)) is plotted against pressure

The following graph is plotted between compressibility factor Z versus pressure of a gas at different temperatures.Which of the following statements is /are correct?
from
per adult (price varies by group size)







