Gas compressibility factor Z: Ideal gas vs Real gas
By A Mystery Man Writer
Description
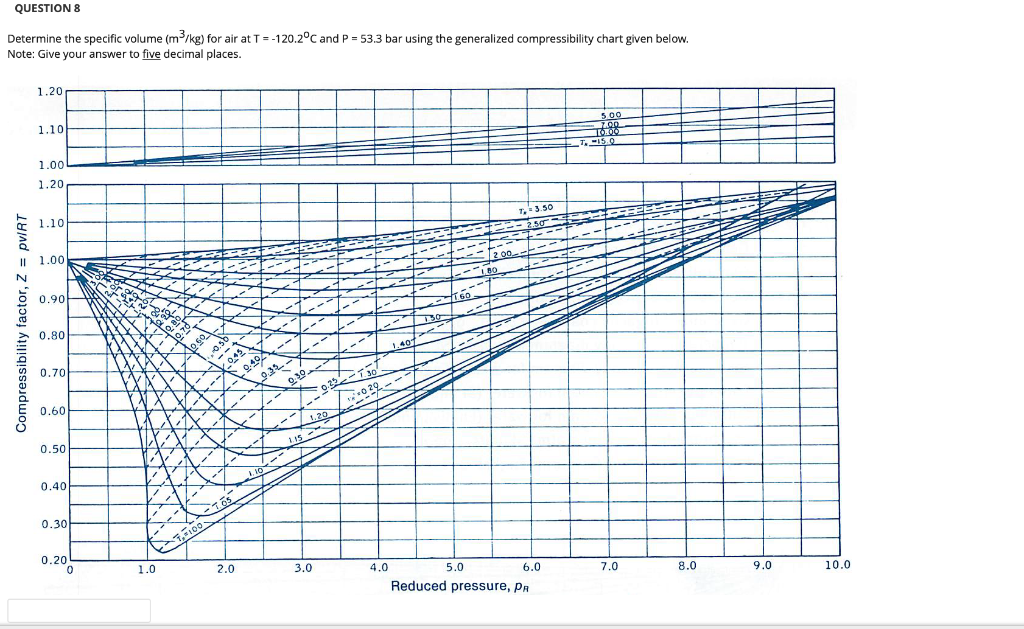
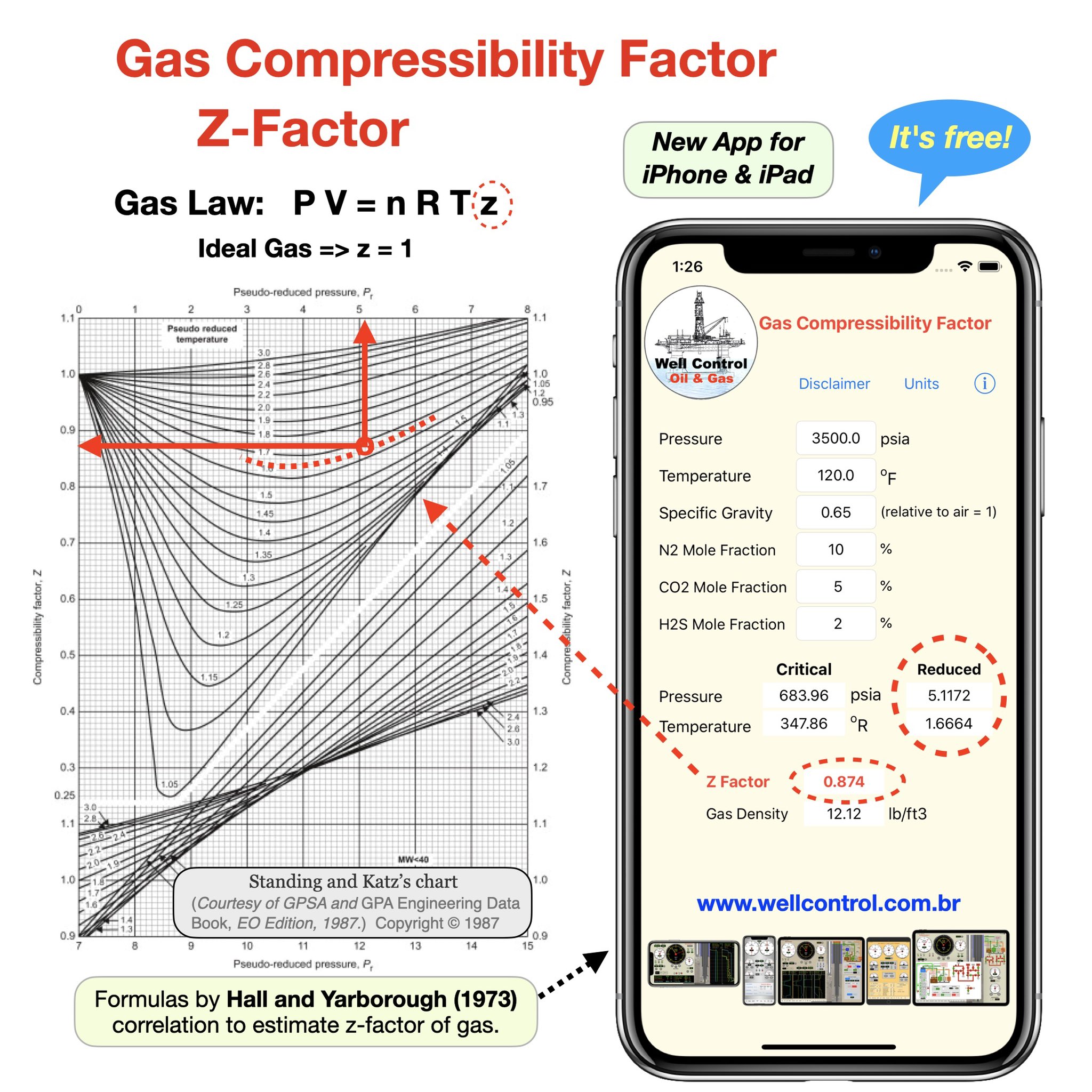
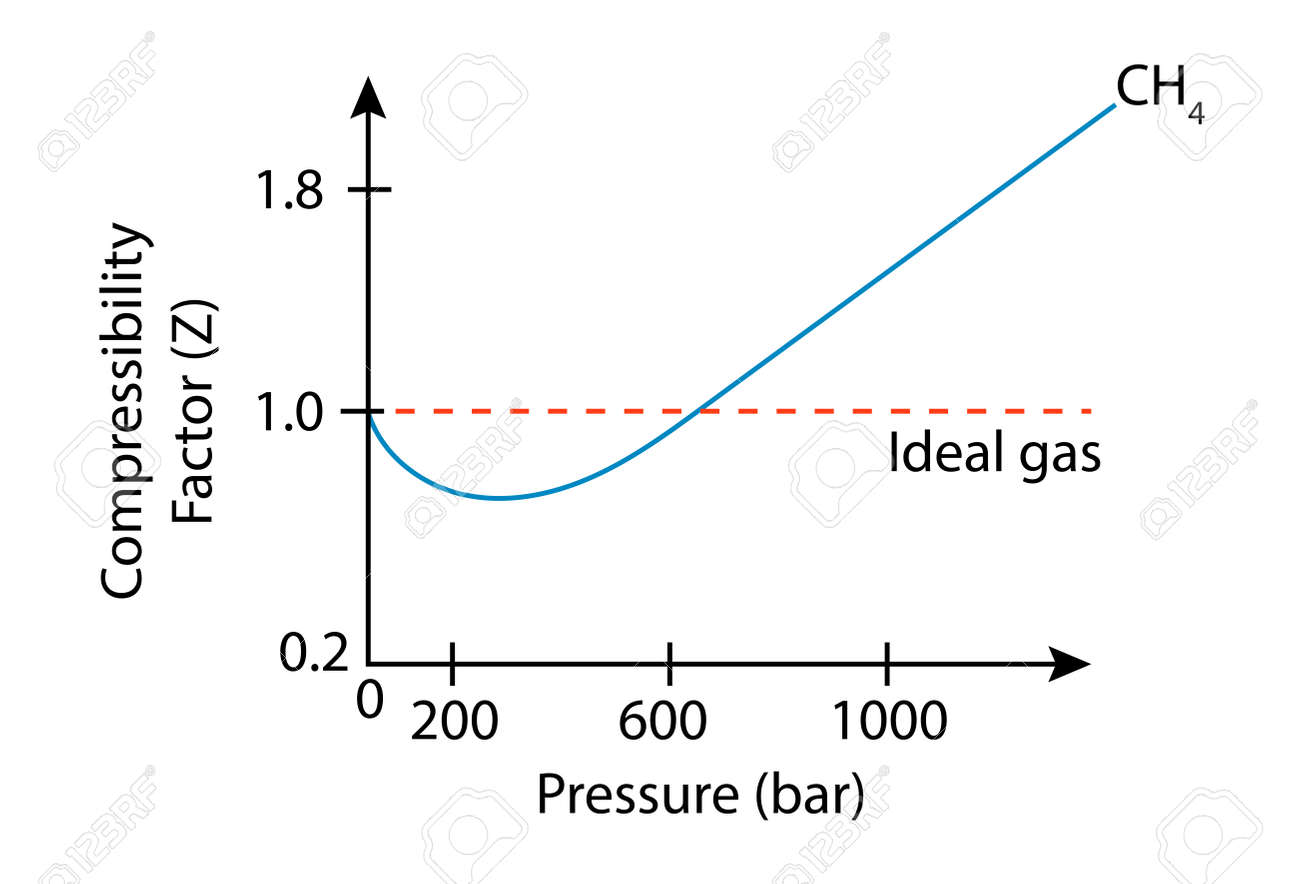
Gas compressibility factor, Z, and Gas compressibility are not the same. Gas compressibility factor Z is the ratio of the gas volume at a given temperature and pressure to the volume the gas would occupy if it were an ideal gas at the same temperature and pressure.

Objectives_template

GOR is not a good indicator of reservoir fluid type in tight liquid-rich shale plays

Pressure Maintenance in the Groningen Gas Field (Part III)

For an ideal gas, the value of compressibility factor `Z(=(pVm)/(RT))` is

Compressibility factor (Z=(PV)/(nRT)) is plotted against pressure

What is the main difference between Process Plant Piping Engineering and Pipeline Engineering.?

Non Ideal Gas Behavior-chemistry - Non Ideal Gas Behavior Calculate the compressibility factor (Z) - Studocu

Select the incorrect statement:aCompressibility factor foran ideal gas is unity.A real gas approachesideal

Non-Ideal Gas Behavior Chemistry: Atoms First

Compressibility factor - Wikipedia

5.13-Real gases, compressibility factor, deviation of real gas from ideal behaviour, state of matter

physical chemistry - Why do some gases have lower value of Z for a particular pressure? - Chemistry Stack Exchange
from
per adult (price varies by group size)