Compression of a gas due to external pressure and the
By A Mystery Man Writer
Description

2 mole of an ideal gas undergoes isothermal compression along three different paths

One mole of an ideal gas is compressed from 500 cm^(3) against a

Atoms/molecules collide with the wall of a piston with area A

PDF) Natural laws and ontological reflections: the textual and

Waldo QUIROZ, Professor (Full), PhD Chemistry

Phase Changes
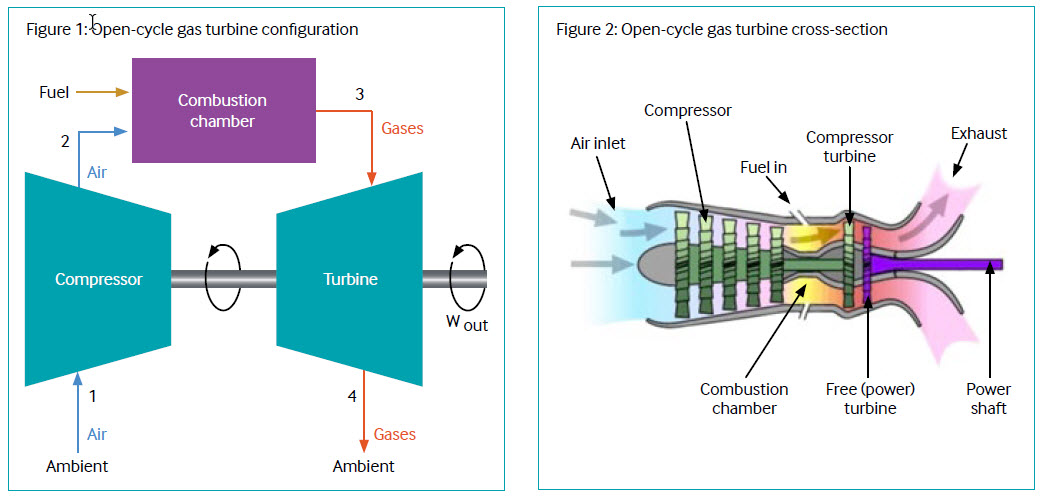
Open-cycle gas turbines (2022)

An ideal gas undergoes isothermal compression from { 5m }^{ 3 } to 1 m^3 against a constant external pressure of 4 N{ m }^{ -2 }. Heat released in this process

PDF) Natural laws and ontological reflections: the textual and

1 (3) 248.5 K Pere R2- (4) 200 K La The work done in adiabatic compression of 2 mole of an ideal monoatomic gas by constant extemal pressure of 2 atm

⏩SOLVED:A gas is compressed from an initial volume of 5.55 L to a…
Two moles of an ideal gas is compressed isothermally and reversibly from a volume 2L to 0.5L at initial pressure of 1 atm . the work done by gas i

Waldo QUIROZ, Professor (Full), PhD Chemistry

PDF) Natural laws and ontological reflections: the textual and

Boyle's Law Clippard Knowledgebase
from
per adult (price varies by group size)


.jpg)



