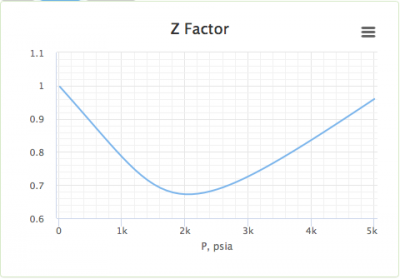
OneClass: For a real gas, the compressibility factor, Z, is
By A Mystery Man Writer
Description

Non-Ideal Gas Behavior Chemistry: Atoms First

OneClass: For a real gas, the compressibility factor, Z, is defined as Z (T, P) = PV/nRT For an ideal

physical chemistry - Is the compressibility factor smaller or greater than 1 at low temperature and high pressure? - Chemistry Stack Exchange
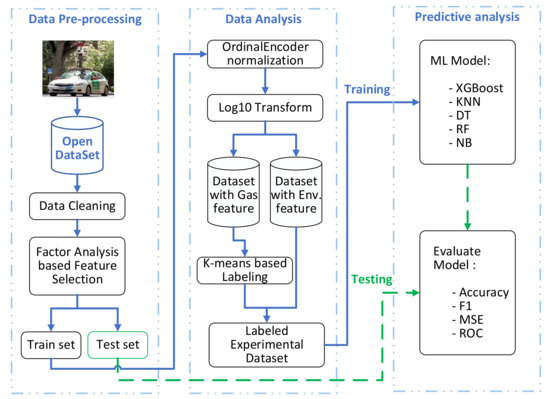
Applied Sciences, Free Full-Text

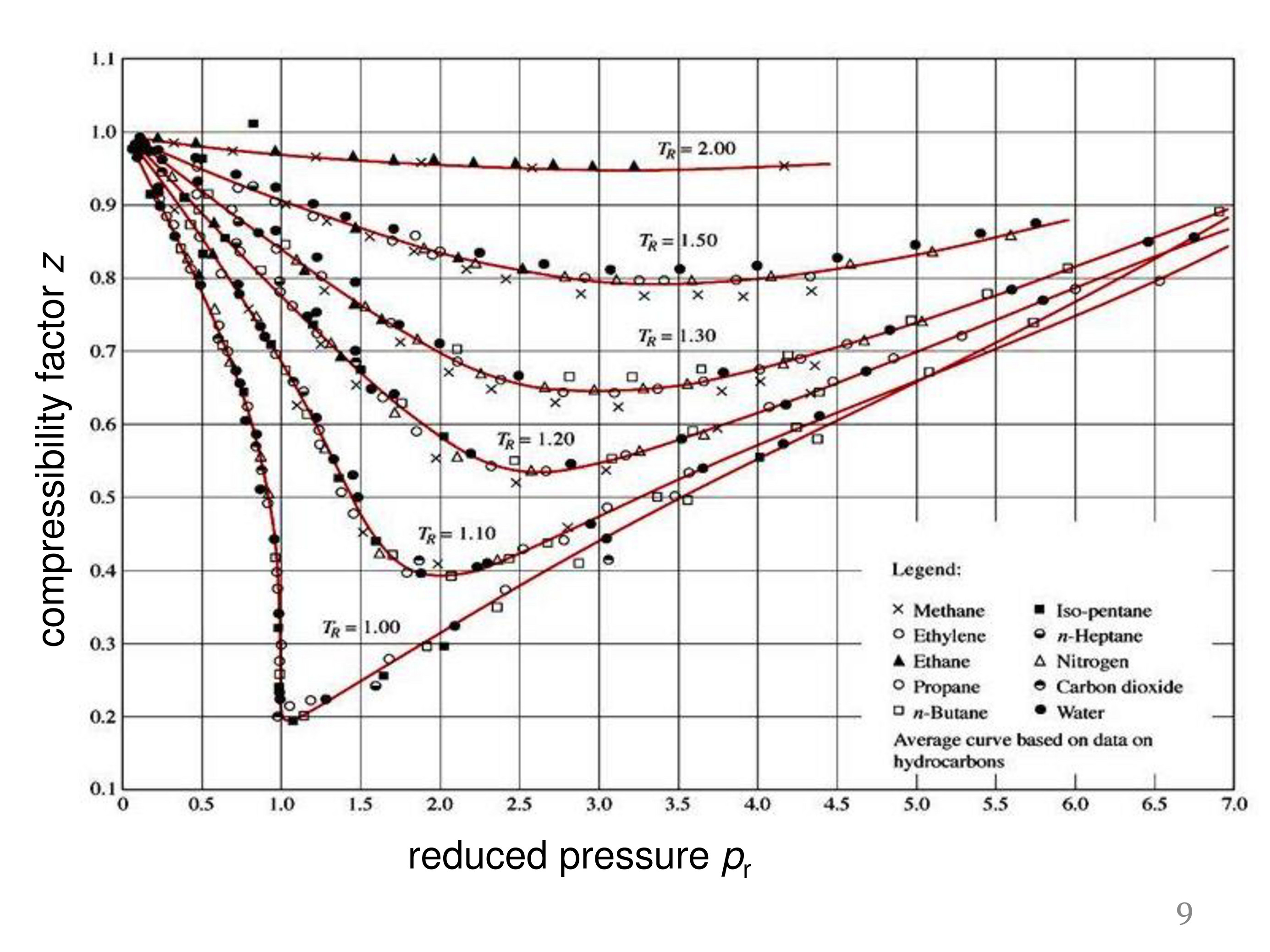
Determine Compressibility of Gases

Energies, Free Full-Text

The compressibility factor Z a low-pressure range of all gases except hydrogen is:Z=(1+ displaystylefrac{a}{V_{m}RT})Z =(1-displaystylefrac{a}{V_{m}RT})Z=(1+displaystylefrac{Pb}{RT})Z = ( 1 - displaystylefrac{Pb}{RT})
Ideal gases and real gases are compressible or not compressible what is the compressible factor for real gases and ideal gases.

OneClass: The higher the pressure of the gas the lower its compressibility! 2. Find K, for a. a van d
States of Matter Class 11 Notes CBSE Chemistry Chapter 5 [PDF]
from
per adult (price varies by group size)