The given graph represent the variations of Z Compressibility
By A Mystery Man Writer
Description
The compressibility factor is actually a factor that corrects the actual value of the gas versus the ideal gas. Let us learn and understand this concept.
What is the compressibility factor, and how does it vary with an increase in temperature and pressure? Watch this video to get the answer. This is an importa
What is the compressibility factor, and how does it vary with an increase in temperature and pressure? Watch this video to get the answer. This is an importa

Gaseous State JEE, PDF, Gases

e Compressibility factor (Z) for hydrogen WRT pressure and temperature

The compressibility factor Z a low-pressure range of all gases except hydrogen is:Z=(1+ displaystylefrac{a}{V_{m}RT})Z=(1-displaystylefrac{a }{V_{m}RT})Z=(1+displaystylefrac{Pb}{RT})Z = ( 1 - displaystylefrac{Pb}{RT})

The given graph represents the variations of compressibility factor Z = , versus P, for three real gases A, B and C. Identify the only incorrected statement

The given graph represents the variation of Z(compressibility
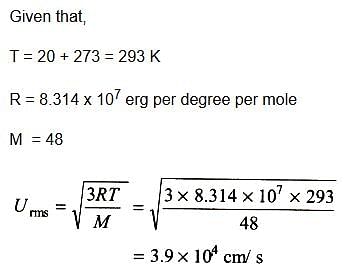
Gaseous State - 3 Free MCQ Practice Test with Solutions - Chemistry
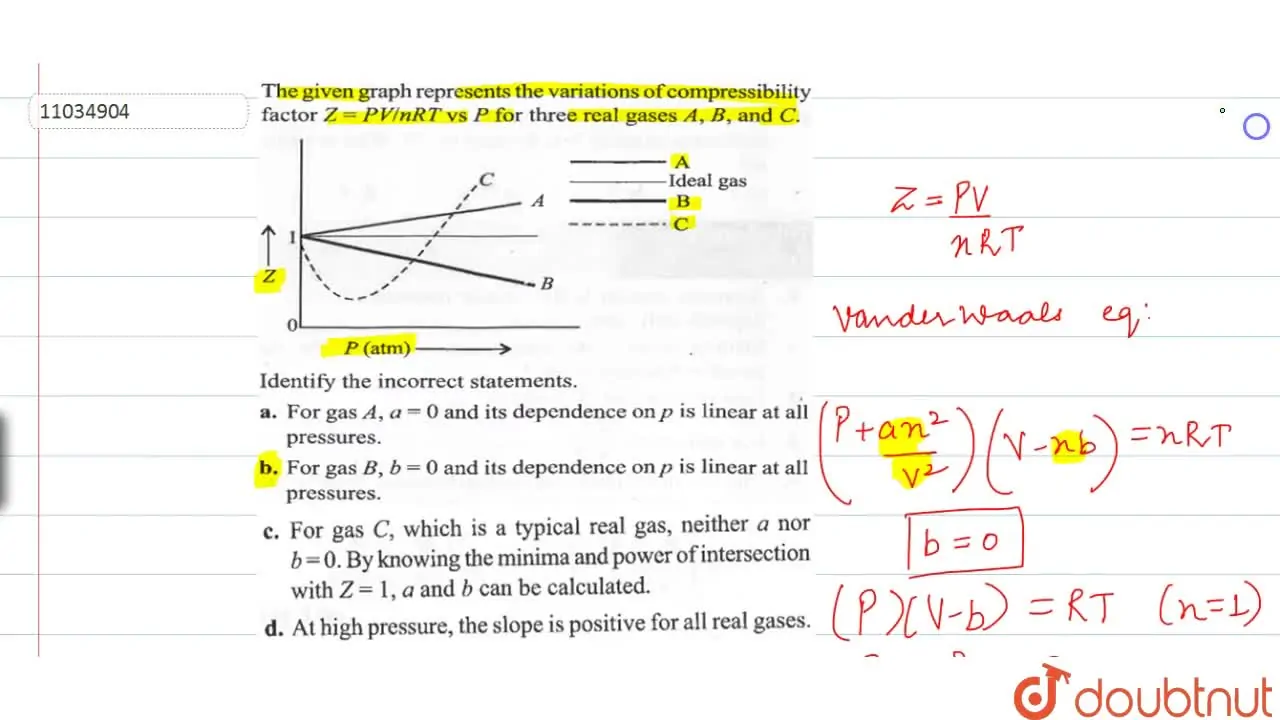
For the gas C which is typical real gas for which neither a nor b=0. B
Negative deviation in PV vs P graph

Determine Compressibility of Gases

Gaseous State Questions for JEE exam - Free Online All questions
from
per adult (price varies by group size)







