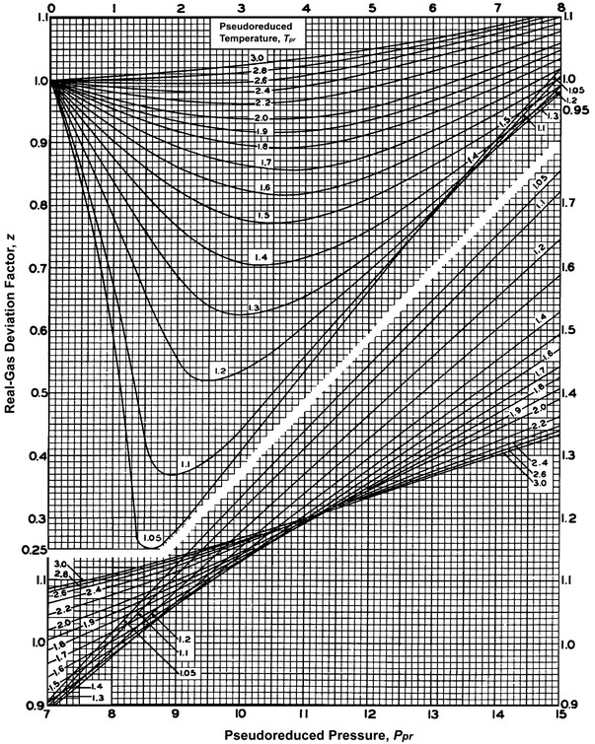
If `Z` is a compressibility factor, van der Waals' equation at low pressure can be written as
By A Mystery Man Writer
Description
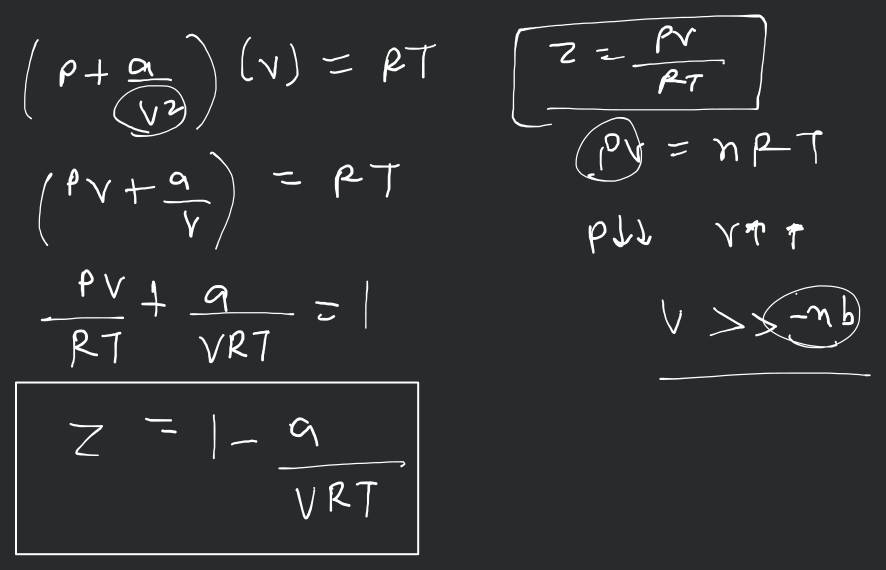
If Z is a compressibility factor, van der Waals equation at low pressure ..

⏩SOLVED:If Z is a compressibility factor, van der Waals equation at…

Given Vapour pressure of H 2 O at 300 K is 3170 Pa R 8314 JK 1 mol 1 2010 A 127

SOLUTION: Dpp 7 gaseous state and chemical energetics - Studypool

Gaseous State Questions for JEE exam - Free Online All questions of Gaseous State - Chapter-wise Questions of JEE

SOLUTION: Dpp 7 gaseous state and chemical energetics - Studypool

If Z is a compressibility factor, van der Waals' equation at low pressure can be written as

JEE Advanced, Mathematics, Study Material

Class Xi States of Matter, PDF, Gases
Solved papers for JEE Main & Advanced JEE Main Solved Paper-2014
from
per adult (price varies by group size)