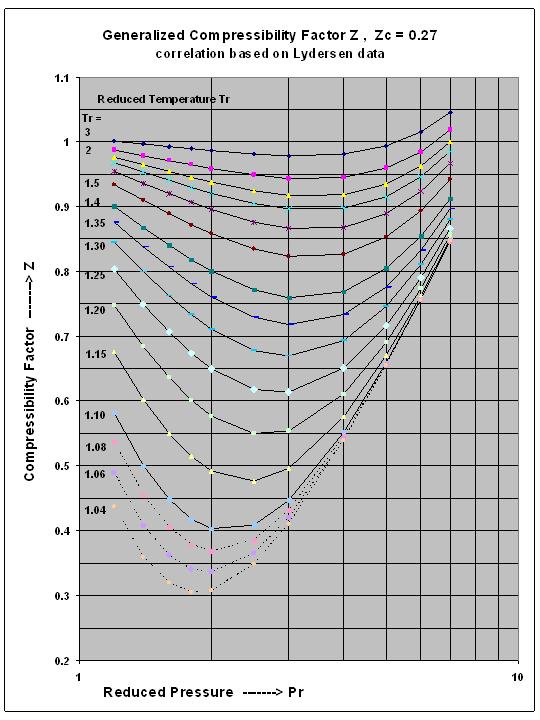
The compressibility factor Z a low-pressure range of all gases
By A Mystery Man Writer
Description
Click here:point_up_2:to get an answer to your question :writing_hand:the compressibility factor z at a lowpressure range of all gases except hydrogen is
Click here👆to get an answer to your question ✍️ The compressibility factor Z a low-pressure range of all gases except hydrogen is-Z-1- displaystylefrac-a-V-m-RT-Z-1-displaystylefrac-a-V-m-RT-Z-1-displaystylefrac-Pb-RT-Z - - 1 - displaystylefrac-Pb-RT-
The van der Waals equation for real gases is -P-aVm2-Vm-x2212-b-RT
Click here👆to get an answer to your question ✍️ The compressibility factor Z a low-pressure range of all gases except hydrogen is-Z-1- displaystylefrac-a-V-m-RT-Z-1-displaystylefrac-a-V-m-RT-Z-1-displaystylefrac-Pb-RT-Z - - 1 - displaystylefrac-Pb-RT-
The van der Waals equation for real gases is -P-aVm2-Vm-x2212-b-RT

Investigation of the Properties of Hydrocarbon Natural Gases Under Confinement in Tight Reservoirs Due to Critical Properties Shift

Compressibility factor Z - Gaseous State

Compressibility factor - Wikipedia

Deviation of Real Gases from Ideal Gas Behaviour - GeeksforGeeks

The role of the compressibility factor Z in describing the volumetric behavior of gases

Compressibility Factor Calculator

Lecture 4-Real-Gases, PDF, Gases

Compressibility Factor Charts - Wolfram Demonstrations Project
How can a gas be ideal at a high pressure and low temperature? - Quora

Chemistry!!! Not Mystery : Do Real Gases Behave Ideally?

Plot of experimental measurements of the z-factor

COMPRESSIBILITY factor Z, Using P and v in 3 Minutes!
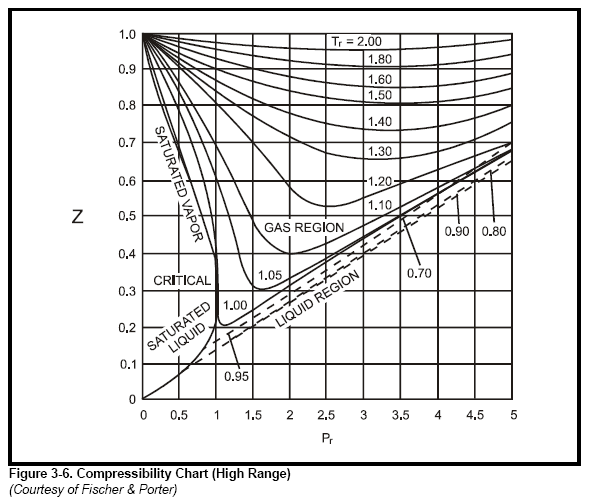
Chapter 3 - Physical Properties of Fluids: Gas Compressibility Factor
from
per adult (price varies by group size)