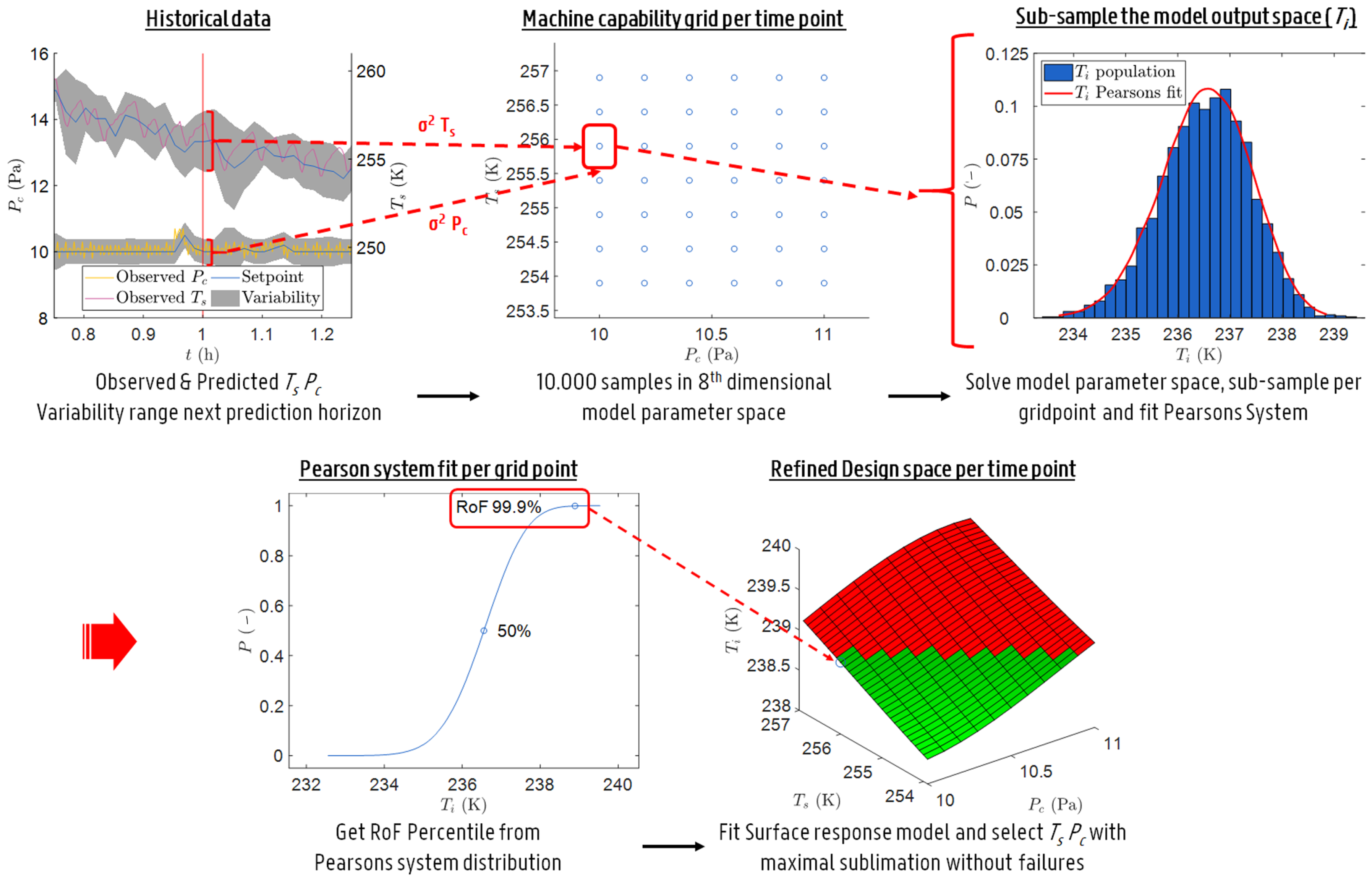
Example of a primary drying design space graph showing sublimation
By A Mystery Man Writer
Description

PDF) Recommended Best Practices for Lyophilization Validation—2021
Pharmaceutics, Free Full-Text

Recommended Best Practices for Lyophilization Validation—2021 Part

Freeze-drying parameters for uncontrolled and controlled
Foods, Free Full-Text

Practical Advice on Scientific Design of Freeze-Drying Process: 2023 Update

Technical transfer and commercialisation of lyophilised biopharmaceuticals — application of lyophiliser characterisation and comparability, AAPS Open

Figure A1. Product temperature for 5.4 mL and 10.6 mL fill volumes

Petr KAZARIN, Postdoctoral Research Associate
from
per adult (price varies by group size)