An ideal gas is taken from (Pi, Vi) to (Pf, Vf) in three different ways. Identify the process in which the work done on the gas the most. - Physics
By A Mystery Man Writer
Description
An ideal gas is taken from (Pi, Vi) to (Pf, Vf) in three different ways. Identify the process in which the work done on the gas the most.

Graphically show the total work done in an expansion when the state of an ideal gas is changed reversibly and isothermally from pi, Vi to pf, Vf With the help of p
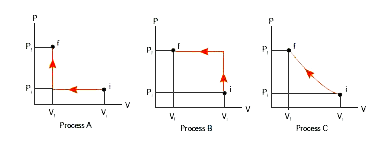
An ideal gas is taken from (P(i),V(i)) to (P(f),V(f)) in three differe

Heat Engines and Efficiency - ppt download

Solved P An ideal gas can be taken from state a to state c

Thermodynamics: An Engineering Approach - 5th Edition - Part I by 黑傑克 - Issuu
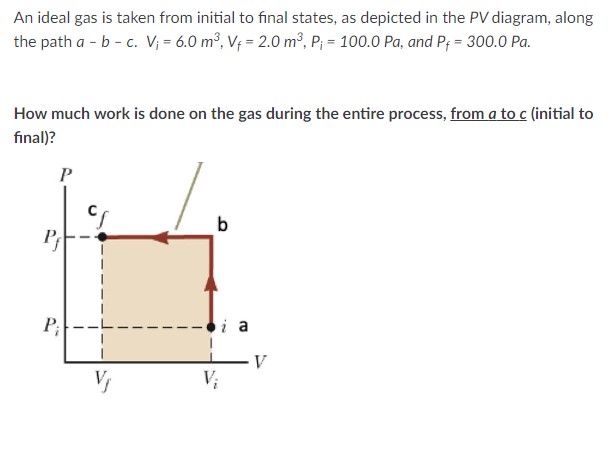
Solved An ideal gas is taken from initial to final states

Physics Web Assign Ch 12 #8

Thermodynamics - Physics at Oregon State University
What are all the applications of an isothermal process? - Quora

Atkins' Physical Chemistry [12 ed.] 9780198847816
Visualizing and characterizing excited states from time-dependent density functional theory - Physical Chemistry Chemical Physics (RSC Publishing) DOI:10.1039/D3CP04226J
Five moles of an ideal gas are compressed isothermally from A to B, as the graph illustrates. What is the work involved if the temperature of the gas is 307 K? Be

Phys 1011 Module AAU, PDF, Euclidean Vector
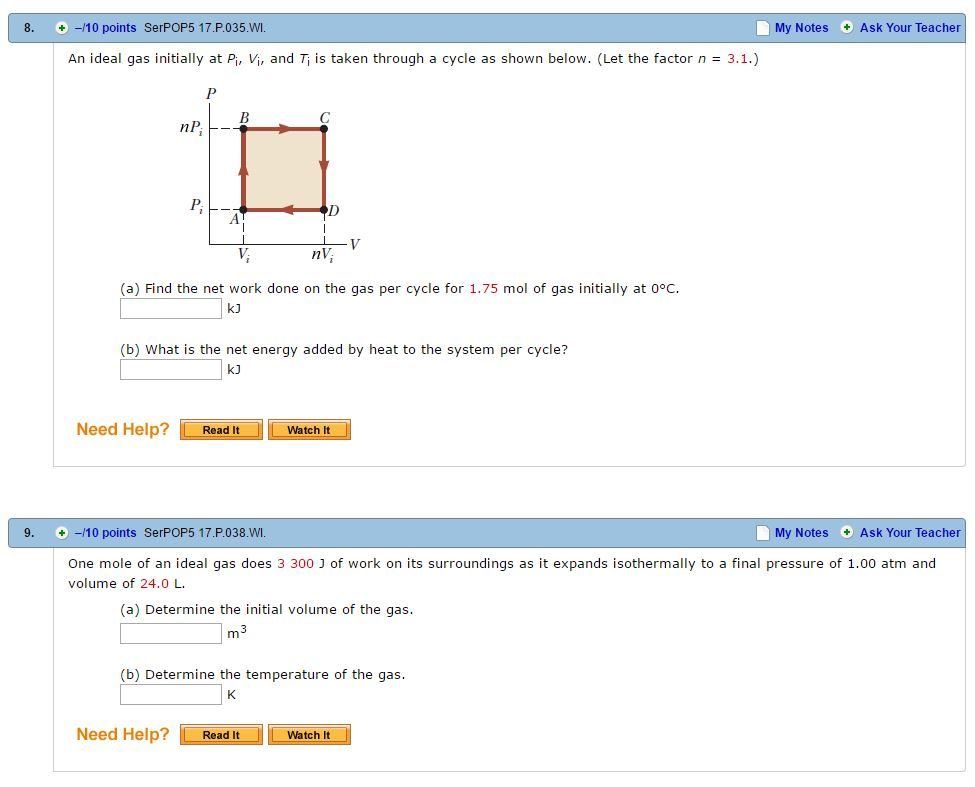
Solved An ideal gas initially at P_i, V_i, and T_i is taken

Heat and Thermodynamics - 2 PDF, PDF, Thermodynamic Equilibrium
from
per adult (price varies by group size)







